Introduction: The Flagship of Somatotropic Targeting
Within the tier of synthetic Growth Hormone-Releasing Hormone (GHRH) analogs, Tesamorelin occupies the apex of the clinical hierarchy. First approved for profound visceral adipose tissue (VAT) reduction in lipodystrophy models (as Egrifta®), it sets the gold standard for specific, non-stimulant visceral fat cleavage.
Unlike truncated fragments (such as AOD-9604) or short-chain secretagogues, Tesamorelin is a massive, full-length 44-amino acid peptide that acts identically to endogenous GHRH. However, due to its complex molecular weight and delicate structural folding, improper laboratory handling rapidly destroys the compound. Establishing precise Tesamorelin reconstitution standards is not arbitrary—it is a strict bio-chemical imperative to prevent molecular denaturation prior to administration.
Section 1: The Master N-Terminal Modification Strategy
Natural, endogenous GHRH possesses a crippling biological flaw when utilized exogenously: it is immediately annihilated by the dipeptidyl peptidase-4 () enzyme in the bloodstream. The biological half-life of natural GHRH is less than 7 minutes—far too short to induce meaningful cellular receptor signaling.
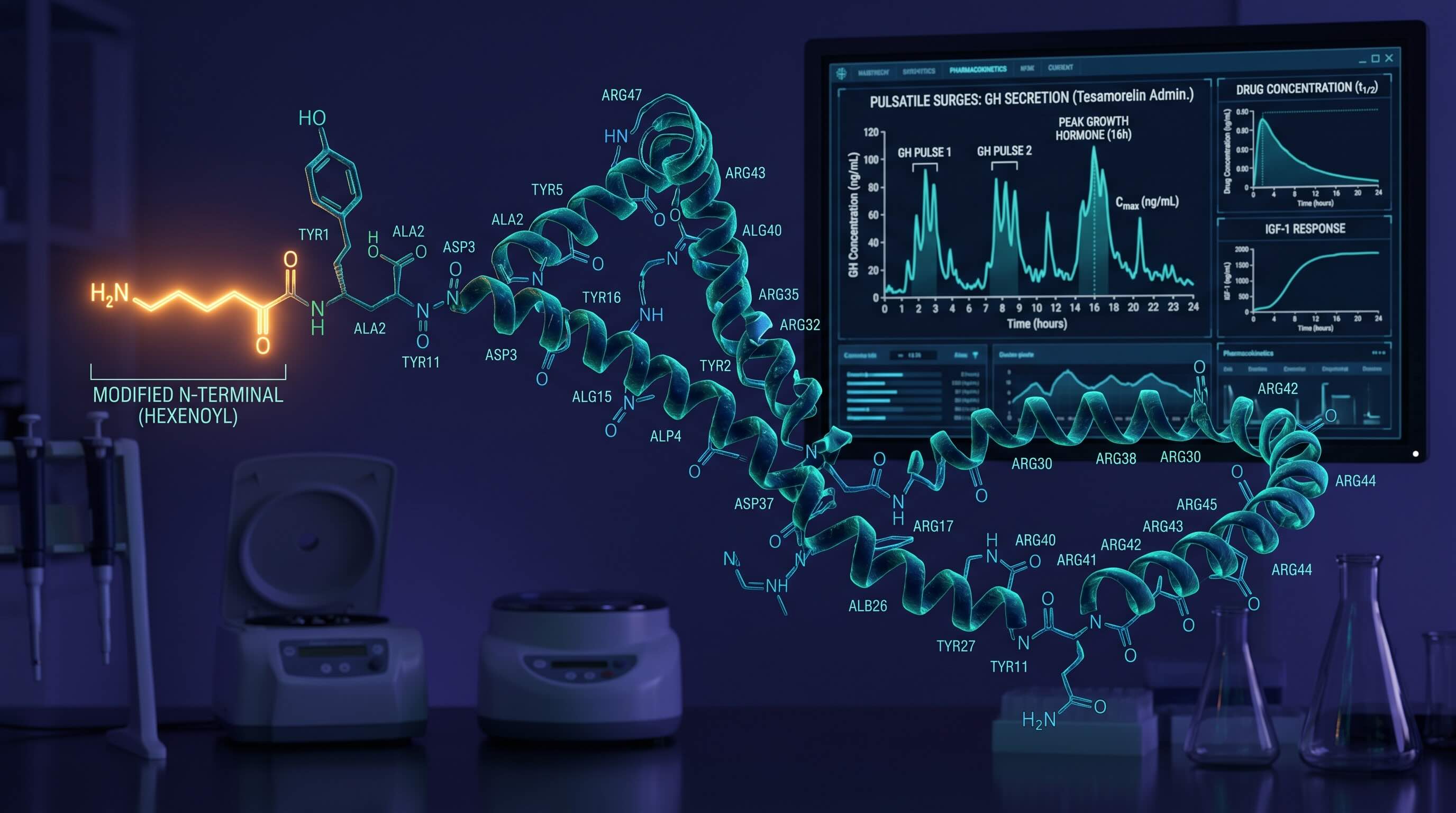
To circumvent this, researchers synthetically engineered Tesamorelin by attaching a highly specific hexenoyl group () to the N-terminal tyrosine residue at position 1.
- Enzymatic Resistance: The trans-3-hexenoic acid attachment acts as a macromolecular shield, rendering the typically vulnerable Tyr-Ala amino sequence completely invisible to cleavage.
- Receptor Saturation: This structural "armor" artificially extends the half-life of Tesamorelin, allowing it to survive in the plasma long enough to travel to the anterior pituitary and latch onto GHRH receptors, stimulating sustained, pulsatile surges of somatropin.
Establishing Steady-State Serum Levels ()
High-performance research indicates that VAT reduction is not accomplished through acute spikes, but through the establishment of a sustained Steady-State Concentration (). Unlike continuous infusion models, Tesamorelin relies on episodic (daily) pulse modeling to mimic natural chronobiology without desensitizing the receptor.
Mathematical Modeling of the Accumulation Envelope
To calculate the steady-state baseline established via repeated daily administration (), we utilize the standard accumulation equation accounting for the drug's elimination constant ():
Because the elimination constant of Tesamorelin clears the vast majority of the exogenous peptide within 6 hours, chronobiological saturation () effectively zeroes out daily. This specific mechanism forces the pituitary to output a massive acute biological GH wave without suppressing endogenous baseline production. A standard to pulse is required to breach the therapeutic threshold.
Synergy Tip: The Somatotropic Amplifier
While Tesamorelin powerfully targets Visceral Adipose Tissue (VAT), adding a GHRP-2 analog can profoundly amplify the total somatotropic output through entirely non-competitive receptor pathways.
Section 2: Precision Tesamorelin Reconstitution Standards
Because Tesamorelin is notoriously sensitive to spatial crowding and solvent acidity, its handling parameters are highly inflexible. The clinical Tesamorelin reconstitution standards differ significantly from smaller BPC-157 or GHRP molecules.
The global baseline for premium research-grade Tesamorelin is the . Unlike standard sequence vials which tolerate deep dilution, the structure must be kept highly concentrated to deliver the required mass without exceeding subcutaneous fluid volume limits.
The 1:1 Calibration Matrix
- 1.Solvent Constraint ():Administering precisely of Sterile Water for Injection (SWFI) or Bacteriostatic Water establishes a perfect 1:1 ratio.
- 2.Density Output ():This density ensures that the standard daily clinical target () requires a spatial draw of exactly 50 Units () or 100 Units ().
- 3.Agitation Ban:Due to the long 44-chain structure, Tesamorelin is exceedingly fragile. Roll the vial between the palms. Never shake.
Section 3: Clinical Trials and Egrifta Documentation
The metabolic supremacy of Tesamorelin is firmly grounded in FDA-approved clinical trials (initially evaluated for HIV-associated lipodystrophy). High-grade clinical data establishes that daily administrations of over a 26-week horizon yielded visceral adipose tissue (VAT) reductions exceeding .
Core Reference Literature
- 1. Falutz J, Allas S, Blot K, et al. Metabolic effects of a growth hormone-releasing factor in patients with HIV. New England Journal of Medicine. 2007;357(23):2359-2370.
- 2. Stanley TL, Falutz J, Marsolais C, et al. Reduction in visceral adiposity is associated with improved lipid profile in HIV-infected patients receiving tesamorelin. Clinical Infectious Diseases. 2012;54(11):1642-1651.
- 3. Egrifta (tesamorelin) Prescribing Information. Theratechnologies Inc.
Handling Imperatives
When working with an analog sequence of this complexity and financial scale, researchers must isolate standard variables. Reconstitute exclusively using the precise Tesamorelin reconstitution standards listed above. If you are integrating Tesamorelin alongside an Ipamorelin stack to create synergistic phase amplitudes, utilize our designated laboratory protocol planner to ensure injections are staged optimally against circadian fasting windows.