Universal Peptide Dosage Calculator: Precision Calibration for Lyophilized Compounds
Calculate precise peptide reconstitution and dosing architecture for U-100 syringes. Professional-grade modeling for clinical research and academic protocol standardizations.
Research Use Only - Important Disclaimer
This tool is for educational and laboratory research purposes only. Not for human consumption. Always consult a licensed medical professional before using any peptides or medications. Improper use can be dangerous.
Peptide Calculator
Calculate precise dosing for U-100 insulin syringes
Input Values
Click unit to toggle between mg and mcg
Concentration
5.00
mg/mlVisual Guide
Calculated Dose
0.00 units
on U-100 Insulin Syringe
Peptide-Specific Protocol Mappers
Pre-configured volumetric matrices with targeted typical dosing intervals for primary research analogs.
Semaglutide
Weight loss & metabolic health peptide calculator
BPC-157
Healing & injury recovery peptide calculator
TB-500
Recovery & flexibility peptide calculator
PT-141
Personal health peptide calculator
CJC-1295 & Ipamorelin
Growth hormone stack calculator
Retatrutide (LY3437943)
Triple G agonist weight loss peptide calculator
Tirzepatide (Mounjaro)
Dual GIP/GLP-1 agonist peptide calculator
Protocol Planner
Create and print a custom 4-week injection schedule for your fridge
Section 1: The Unified Field of Peptide Calibration
The mechanical extraction of a lyophilized compound from a solvent suspension is universally governed by absolute physical mass thresholds. Irrespective of whether the peptide is a sequence of 15 amino acids (like BPC-157) or a massive 43-chain sequence (like TB-500), the underlying algebraic mapping remains flawlessly constant:
Syringe Mechanics: It is imperative for researchers to recognize that a "Unit" on a Standard U-100 syringe is exclusively a volumetric measure (). It is never a fixed measure of active mass. The mass transferred inside a Unit solely depends on the mathematical density () established during the initial reconstitution phase.
Section 2: Plunger Parallax and Volumetric Resolution
A prevalent mathematical error in laboratory protocols involves hyper-condensing lyophilized compounds. Attempting to reconstitute a heavy puck with a minimal diluent vector generates an overwhelmingly dense liquid matrix ().
When mapping small titrations against a hyper-dense solution, researchers confront extreme "Plunger Parallax Error." At a density, extracting exactly forces a physical draw of merely (). At this microscopic scale, a visual misalignment of just a single syringe tick-mark instantly alters the dosage increment by a catastrophic . Consequently, modern pharmacological protocols establish a clear "Gold Standard" of deploying or bacteriostatic matrices to purposefully broaden the volumetric scale, expanding into a vastly more manageable or mechanical pull.
Section 3: Step-by-Step Reconstitution SOP
Execute the following formalized sequence to completely negate calculation decay and secure absolute phase stability prior to administration:
- 1.Mass Identification (): Log the exact milligram yield encoded on the structural vial.
- 2.Solvent Integration (): Determine the precise milliliter expansion vector of bacteriostatic water to be injected into the vacuum.
- 3.Density Resolution (): Run the formula to establish the exact liquid density ratio per milliliter.
- 4.Dose Mapping: Deploy this Universal Calculator to align your desired milligram () or microgram () extraction against the physical U-100 syringe ticks.
- 5.Thermal Lock: Immediately transfer the saturated matrix into a strictly temperature-controlled vault.
Section 4: Stability & Handling Excellence
Amino acid chains are exquisitely vulnerable to structural folding deviations when stripped of their protective lyophilized vacuum. Managing spatial handling prevents accidental peptide fracturing prior to cycle administration.
UV Protection
Photodegradation instantly targets carbon bonds. Maintain heavy light-isolation.
Zero Agitation
Never shake the vial. Gravitational swirling is the only authorized kinetic motion.
Lipid Cloudiness
If the reconstituted fluid exhibits particulate cloudiness or fails to dissolve, molecular precipitation has occurred. Discard immediately.
Section 5: Peer-Reviewed Lab Manual References
- Allen LV. The Art, Science, and Technology of Pharmaceutical Compounding. 6th ed. American Pharmacists Association; 2020.
- Akers MJ. Sterile Drug Products: Formulation, Packaging, Manufacturing, and Quality. Informa Healthcare; 2010.
- Gudeman J, Jozwiakowski M, Chollet J, Randell M. Potential risks of pharmacy compounding. Drugs R D. 2013;13(1):1-8.
- Wang W. Instability, stabilization, and formulation of liquid protein pharmaceuticals. International Journal of Pharmaceutics. 1999;185(2):129-188.
Research Insights & Peptide Guides

Mastering Peptide Reconstitution: A Mathematical SOP
Establish volumetric precision and neutralize plunger parallax error using algebraic dose dilution constants.

AOD-9604 Dosing Protocol: Lipolytic Analysis
Establishing a precise protocol by exploring beta-3 adrenergic lipid targeting and IGF-1 independence.

Bacteriostatic Water vs Sterile Water
Understand the biological necessity of bacteriostatic water. Explore 0.9% benzyl alcohol pH buffering and resolving aqueous aggregation.
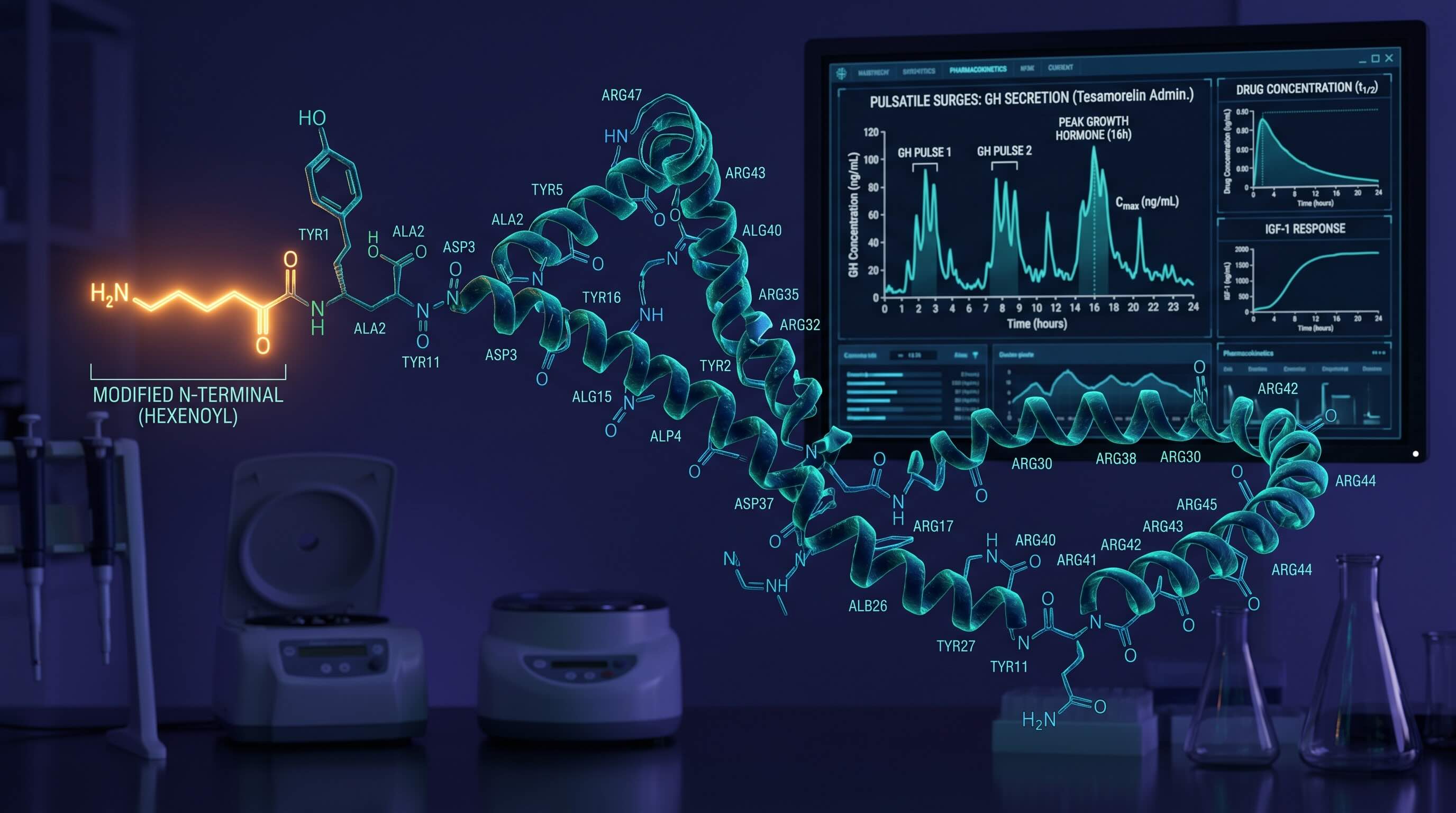
Tesamorelin: Clinical Calibration of GHRH Analogs
Navigating 44-amino acid sequences. Establish authoritative Tesamorelin reconstitution standards to preserve the N-terminal hexenoyl modification.
GHK-Cu Reconstitution Guide: PIP Mitigation
Analyzing the complex tripeptide-copper matrix, pH solubility thresholds, and preventing Post-Injection Pain (PIP).

GHRP-6 vs GHRP-2: Comparative Pharmacodynamics
Analyzing the chemical receptor binding affinity between the two Ghrelin agonists and their resulting GH spike amplitude.